Why do only three companies worldwide manufacture hydrogen-added DEHCH at commercial scale? The answer lies in the process itself. Converting a phthalate ester into a cyclohexanoate ester demands catalyst engineering, high-pressure reactor design, and quality controls that most producers cannot replicate without years of dedicated R&D.
DEHCH — di(2-ethylhexyl) cyclohexane-1,2-dicarboxylate — reaches the market through two distinct production routes, and the route a supplier chooses directly affects product purity, residual phthalate risk, and regulatory eligibility. The molecular structure explains why: saturating the aromatic ring of a phthalate diester eliminates the toxicological concern, but how that saturation happens determines everything downstream.
Raw Materials and Feedstock Chemistry
Both DEHCH production routes start from 2-ethylhexanol (also called isooctanol), the C8 branched alcohol that gives the plasticizer its “2-ethylhexyl” designation. Where the routes diverge is the acid component.
The hydrogenation route begins with phthalic anhydride (PA). PA reacts with 2-ethylhexanol to form DEHP — di(2-ethylhexyl) phthalate — a commodity phthalate plasticizer produced at millions of tons per year globally. The DEHP then enters the hydrogenation reactor. This route leverages cheap, abundant feedstock.
The direct esterification route skips phthalic anhydride entirely. It starts with hexahydrophthalic anhydride (HHPA), a cyclohexane-based acid anhydride where the ring is already saturated. HHPA reacts directly with 2-ethylhexanol to form DEHCH in a single esterification step — no hydrogenation required. The trade-off: HHPA costs more and has fewer suppliers than PA.
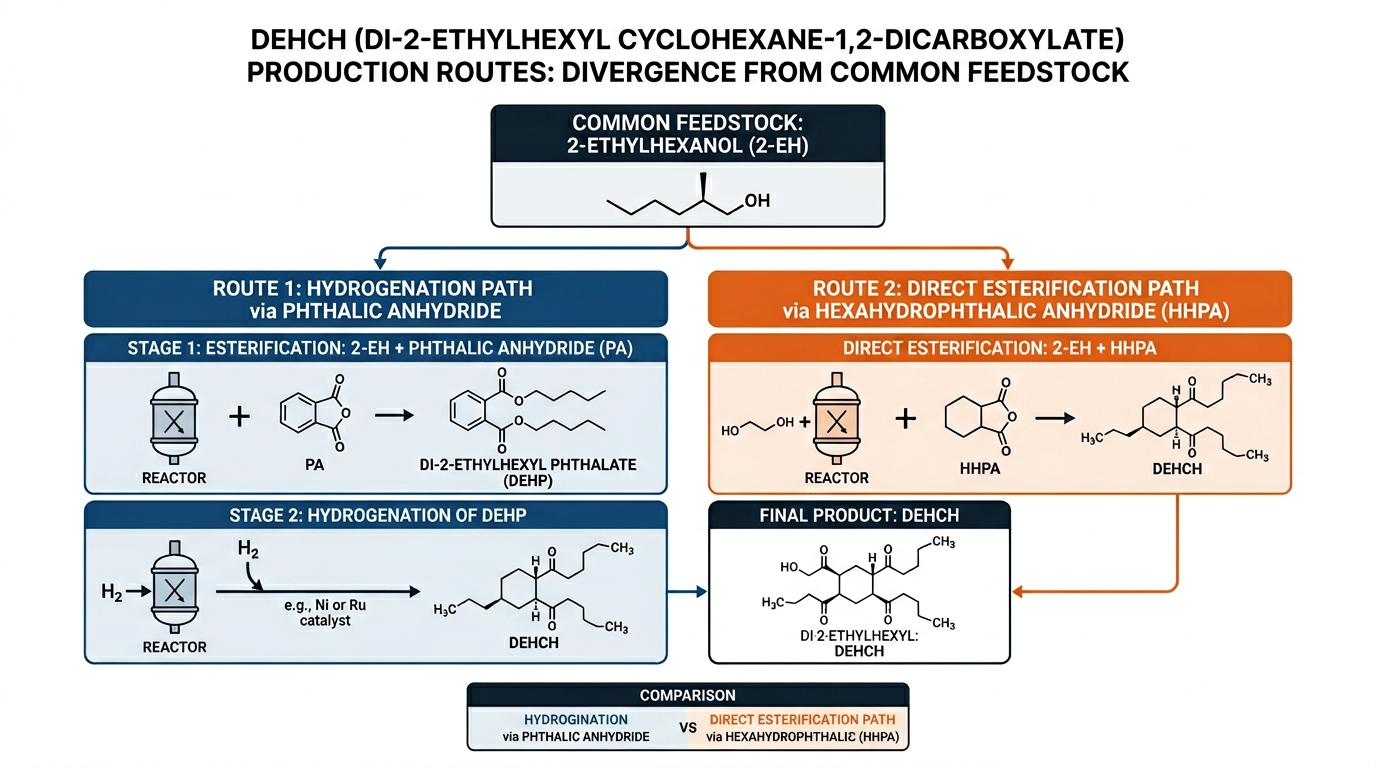
The Hydrogenation Route
The hydrogenation route accounts for the majority of commercial DEHCH production today. BASF, Evonik, and Hanwha Solutions are the three companies that have commercialized this technology, with Hanwha investing eight years of R&D before reaching production at their Ulsan facility.
Esterification of the Phthalate Precursor
Phthalic anhydride and 2-ethylhexanol react at 160-220C under acid catalyst (typically titanium alkoxide or p-toluenesulfonic acid) to form DEHP. This esterification is the same process that produces the DOP plasticizer used worldwide. Target acid number at this stage: below 0.1 mg KOH/g. Any residual free acid or alcohol carries into the hydrogenation reactor and causes problems there.

Catalytic Hydrogenation of the Aromatic Ring
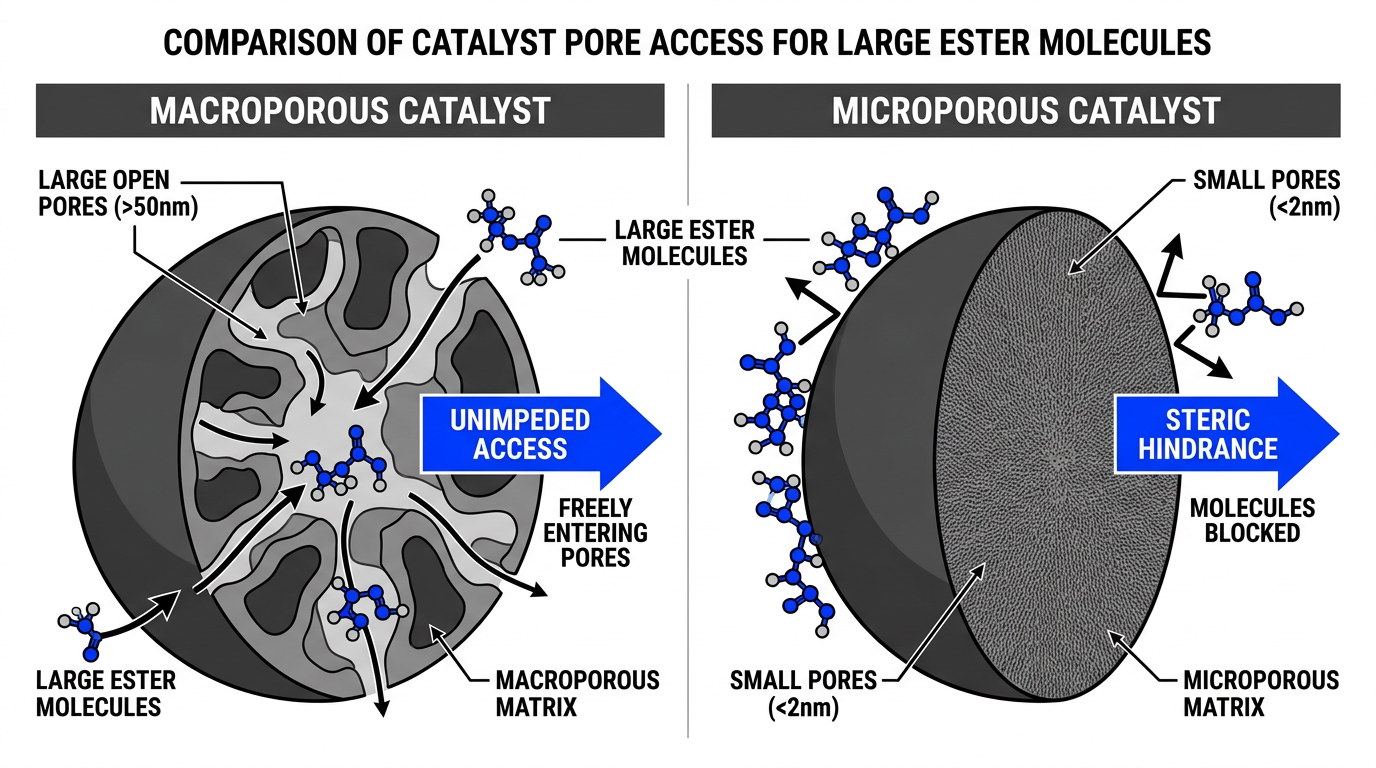
The DEHP feedstock enters a high-pressure reactor charged with hydrogen gas and a supported noble metal catalyst at 70-220C and 20-300 bar. Ruthenium or palladium on macroporous alumina supports are the standard choices.
The counterintuitive insight in catalyst selection: conventional high-surface-area catalyst supports actually underperform here. DEHP molecules are large (C24 backbone), and they cannot diffuse into micropores or mesopores efficiently. Macroporous supports with pore diameters of 50-10,000 nm and BET surface area below 30 m2/g give the bulky ester molecules full access to the active metal sites throughout the particle. The result is 100% conversion with 99.2% yield at 120C and 200 bar — benchmarks demonstrated in continuous operation at space-time yields of 0.1-1 kg per liter of catalyst per hour.
Viscosity management is the other critical factor most process descriptions omit. DEHP feedstock above 20 cP creates stagnant boundary layers around catalyst particles, starving them of dissolved hydrogen. Reducing the liquid-phase viscosity to 0.2-10 cP before reactor entry — through preheating or by co-adding 5-60 wt% 2-ethylhexanol — improves conversion by 20% or more and extends catalyst life. The same 2-ethylhexanol used as a raw material doubles as a process aid: it thins the feedstock and scavenges trace metal ion impurities that would otherwise poison the catalyst.
Purification and Finishing
After hydrogenation, the crude product undergoes vacuum distillation to remove excess alcohol and light ends, followed by neutralization washing and decolorization. The finishing stage targets three critical quality parameters: acid number below 0.05 mg KOH/g (ASTM D974), color index below 20 APHA (ASTM D1209), and GC purity above 99.5%.
Residual phthalate content is the parameter unique to the hydrogenation route. Because the process starts from DEHP, any unconverted feedstock remains as a phthalate impurity. Modern processes achieve residual DEHP below 100 ppm by GC, but this number depends entirely on conversion efficiency — and that ties back to catalyst condition and reactor management.

The Direct Esterification Route
The alternative production path eliminates the hydrogenation reactor entirely. HHPA and 2-ethylhexanol react at a molar ratio of approximately 1.0:2.4, catalyzed by supported methanesulfonic acid at around 2% of total reactant weight. Reaction temperature: 160-180C for 2.5 hours, achieving an esterification ratio of 99.82%.
The equipment is simpler: a standard stirred-tank reactor with reflux condenser, no high-pressure vessel, no hydrogen supply infrastructure, no noble metal catalysts to manage and regenerate. For a producer evaluating capital expenditure, this route has obvious appeal.
The constraint is upstream. HHPA must be produced separately by hydrogenating phthalic anhydride — so the hydrogenation step does not disappear; it moves earlier in the supply chain and produces a different intermediate. HHPA availability varies by region, and any disruption in HHPA supply directly limits DEHCH output. Producers on this route carry feedstock risk that the hydrogenation route avoids by starting from commodity PA.

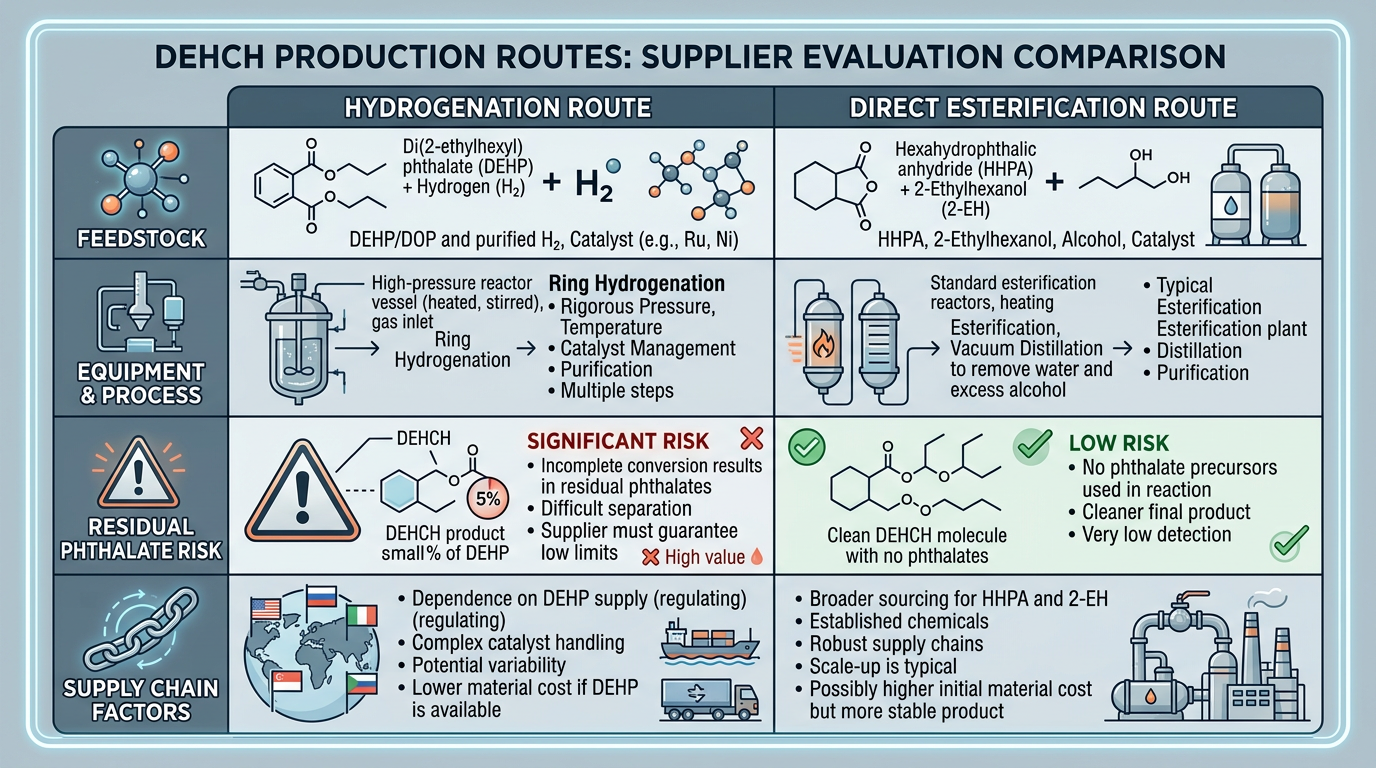
Hydrogenation vs Direct Esterification
The route a supplier uses is not just an engineering preference. It affects the product you receive.
The most consequential difference is residual phthalate content. The hydrogenation route starts from a phthalate ester, so incomplete ring saturation leaves DEHP traces in the final product. At conversion rates above 99.5%, residual phthalate drops below 100 ppm — acceptable for most applications but potentially disqualifying for the strictest food-contact approvals under EU Food Contact Materials regulation or FDA 21 CFR. The direct esterification route never introduces a phthalate compound at any stage. Residual phthalate by GC: effectively zero.
Equipment complexity splits the other direction. The hydrogenation route requires high-pressure reactors rated for 200+ bar, hydrogen gas handling systems, and noble metal catalyst inventory worth hundreds of thousands of dollars. But the feedstock — phthalic anhydride — is one of the cheapest and most available aromatic acids in the world. The direct esterification route needs only conventional atmospheric or low-pressure reactors, but depends on HHPA, a specialty chemical with limited global production capacity.
When evaluating a DEHCH supplier, ask which route they operate. A hydrogenation-route producer should provide GC data for residual phthalate content on every COA. A direct esterification-route producer should demonstrate consistent HHPA sourcing. Both should report acid number, color index, and purity by GC — but the residual phthalate line item tells you immediately which process produced the material.
Claims that one route costs less than the other deserve scrutiny. The hydrogenation route adds hydrogen cost and catalyst overhead; the direct esterification route pays a premium for HHPA feedstock. Without knowing a producer’s specific catalyst lifecycle and HHPA procurement terms, blanket cost comparisons between the two cyclohexanoate routes are unreliable.
Key Takeaways
DEHCH production is not one process but two, and the route determines which quality risks to manage. A COA that omits residual phthalate data from a hydrogenation-route supplier is hiding the single clearest indicator of conversion quality.
The deeper lesson for anyone specifying DEHCH: manufacturing quality is only half the equation. Even perfectly produced DEHCH requires reformulation when replacing a phthalate plasticizer — different gelation behavior, different migration rate, different compatibility window with the polymer matrix. Matching the plasticizer to the formulation is as critical as matching the process to the specification.