DOP datasheets routinely claim “good heat stability.” That claim omits the most critical variable: the stabilizer system paired with it. DOP-plasticized PVC begins yellowing at roughly 70 minutes under 180 C oven aging when the stabilizer disperses poorly in the plasticizer matrix — yet the same resin base retains whiteness past 180 minutes with a different plasticizer that dissolves the stabilizer at twice the concentration. The molecular structure explains why: DOP’s solvating power varies by stabilizer chemistry, and that single variable determines whether a formulation performs for years or fails on the production line.
Calcium-zinc systems compound DOP’s own lubricating effect. Organotin variants disperse readily but need synergistic blending to reach full potential. The compatibility between plasticizer and polymer is never just a binary question — it is a system interaction that shifts with every ratio change.
Calcium-Zinc Stabilizers and DOP: The Double-Lubrication Problem
Ca/Zn stabilizers are metal carboxylates — soaps, in plain chemical terms. Every soap molecule carries inherent lubricity. In a DOP-plasticized compound, that lubricity stacks on top of DOP’s own plasticizing action, creating a double-lubrication effect that narrows the processing window to a point where many formulators struggle to maintain consistent output.
Why Ca/Zn Soaps Compound DOP’s Lubricating Effect
The fatty acid chain length determines how severe the problem gets. Stearates (C18) cause excessive lubrication and delayed fusion. Laurates (C12) offer moderate lubricity but can plate out on processing equipment. When processors switching from lead stabilizers increased Ca/Zn dosage to compensate for lower stabilizing power, the results were predictable: yellow and black lines during extrusion, color instability between batches, and impact strength loss from incomplete fusion.
I have reformulated dozens of flexible PVC compounds through the lead-to-Ca/Zn transition. The pattern is consistent — the processing failures come from treating Ca/Zn as a drop-in replacement when it requires complete system rebalancing including lubricant packages, co-stabilizer additions (phosphites, beta-diketones, epoxy compounds), and tighter temperature control. Without that rebalancing, the double-lubrication effect produces waviness, poor surface finish, and the color instability that frustrates every production shift.

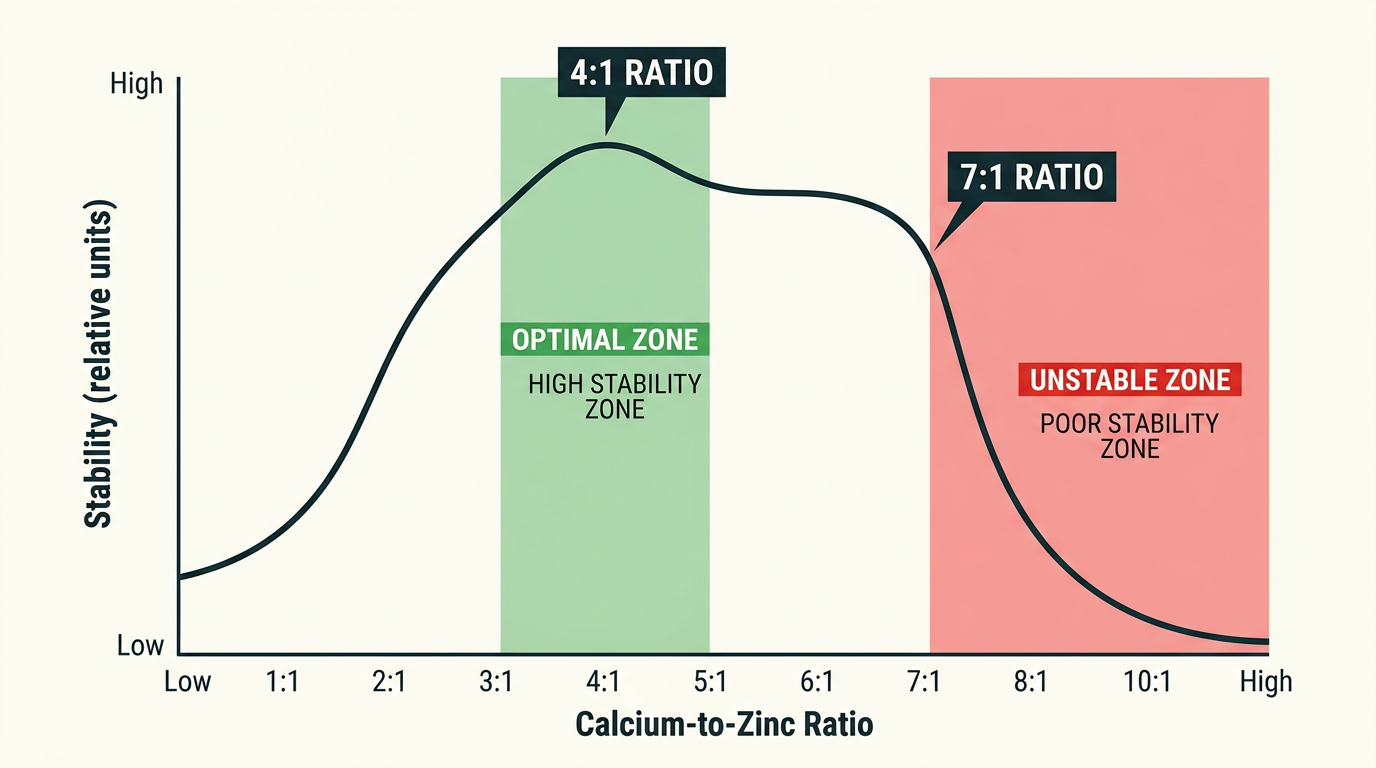
Ratio Optimization and Dosage Limits
A 4:1 calcium-to-zinc molar ratio produces the greatest thermal stability, measured at 15.8 minutes on Congo red at 180 C. Push that ratio to 7:1 and stability drops — excess calcium actually accelerates degradation rather than extending protection.
For DOP-plasticized flexible PVC at standard 40-50 phr DOP loading, keep total Ca/Zn at 3-5 phr and resist the urge to increase dosage when initial trials show early discoloration. The problem is almost never insufficient stabilizer quantity. It is either ratio imbalance or poor dispersion. Adding a polyol co-stabilizer like pentaerythritol doubles thermal stability time versus the Ca/Zn system alone, without worsening the lubrication balance — a far better path than simply adding more soap.
Organotin Stabilizers in DOP-Plasticized PVC
Organotin stabilizers disperse more readily in DOP than Ca/Zn systems. The practical difference is immediate: organotin compounds contribute essentially no lubricity, which means they do not compound DOP’s plasticizing effect. This makes the processing window wider and more forgiving in DOP-plasticized formulations.
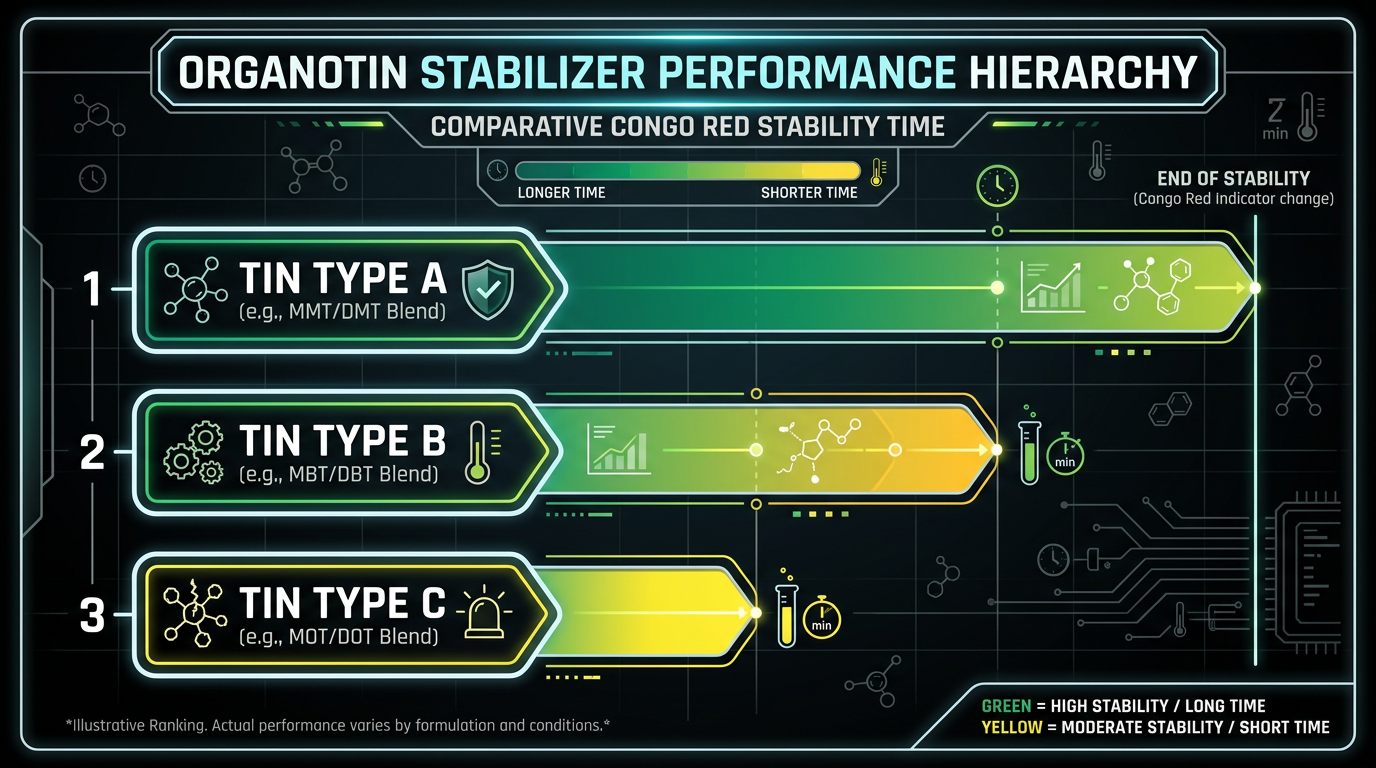
Performance Hierarchy Among Tin Variants
Not all organotins perform equally with DOP. Congo red testing reveals a clear hierarchy among tin neodecanoate variants: dioctyl tin (DOTDN) achieves an initial stability time of 50 minutes, dibutyl tin (DBTDN) reaches 37 minutes, and dimethyl tin (DMTDN) manages only 22 minutes. The performance gap traces directly to viscosity — DMTDN runs at 2,244 mPa-s versus DOTDN at just 218 mPa-s. Lower viscosity means better dispersion in the DOP matrix, and better dispersion means more stabilizer molecules reach reactive sites on PVC chains before degradation begins.
One point that deserves attention: the difference in static thermal stability between DOP and alternative plasticizers like DOA when paired with the same organotin stabilizer does not exceed 4 minutes. Organotin chemistry is far less sensitive to plasticizer choice than zinc-based systems. If your application demands organotin anyway — rigid clear PVC, for instance — the DOP interaction is straightforward.
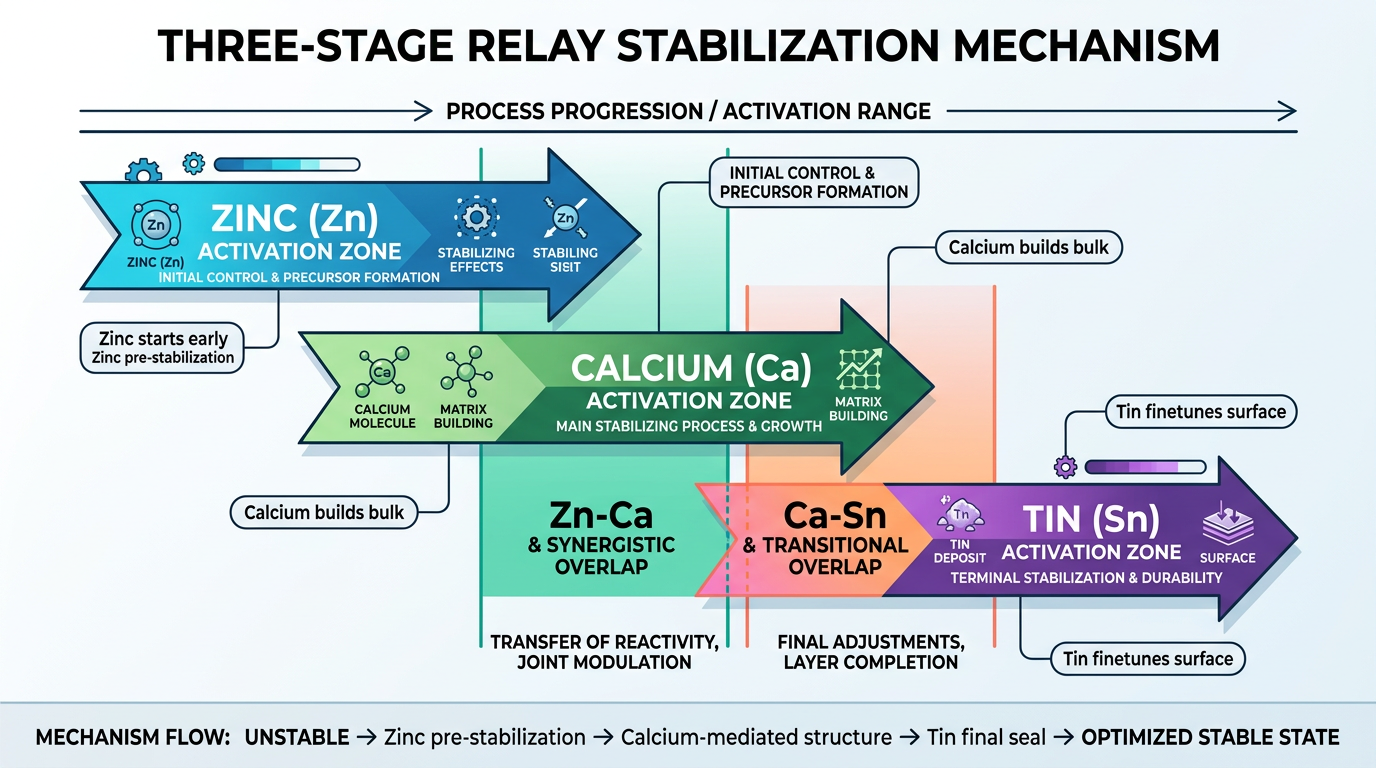
The Organotin-Ca/Zn Relay Mechanism
The most effective stabilizer approach for demanding DOP-plasticized applications combines organotin with Ca/Zn in a synergistic blend. A 5:4:1 ratio of organotin to calcium to zinc (designated OTN5-C4Z1 in published test data) outperforms any single-component stabilizer: initial stability at 55 minutes, secondary stability at 63 minutes, and complete stability at 78 minutes on Congo red.
The mechanism works as a relay. Zinc stearate captures the first wave of HCl released during thermal degradation. Calcium stearate extends the buffer capacity as zinc depletes. Tin mercaptides maintain color by substituting labile chlorine atoms on the PVC backbone. The three components cover successive degradation stages, and the combined result exceeds the sum of individual contributions by 10-15% on Congo red. For high-performance flexible PVC with DOP — cable insulation, medical tubing, automotive interiors — this relay approach gives the widest processing window and longest service life.
Why Identical Formulations Fail Differently
Two batches with the same formulation sheet can produce completely different thermal stability results. The lab says the recipe is correct. The production floor says the product is failing. The disconnect comes from a variable that never appears on a formulation card: stabilizer availability versus stabilizer presence.
DOP dissolves zinc arginine stabilizer at only 0.40 g per 10 mL — less than half the 0.85 g per 10 mL achieved by tributyl citrate. That solubility gap means nearly half the stabilizer sits undissolved in the DOP matrix, unable to reach PVC chain sites where degradation initiates. The stabilizer is present in the formulation. It is not available for protection. This distinction explains field failures that no amount of formulation adjustment resolves, because the problem is physical dispersion, not chemical dosage.
Processing parameters control dispersion quality more than most formulators appreciate. Mixer discharge temperature between 120-130 C enables metallic soaps and epoxides to melt and distribute uniformly through the DOP-PVC matrix. Below 115 C, gelation is incomplete and fish eyes appear — unplasticized particles that signal stabilizer starvation zones. Above 140 C, volatile stabilizer components vaporize before they can protect the resin. That 25-degree window is where consistent thermal stability lives or dies.
I have traced hundreds of “formulation failures” back to processing inconsistency. The fish eyes and powder lumps that trigger batch rejections are almost always dispersion problems, not formulation problems. Before reformulating a DOP-stabilized compound, check the mixer discharge temperature log first. A 10-degree drift explains more failures than any wrong ingredient ratio.

Matching the System, Not Just the Components
DOP compatibility with heat stabilizers is not a property of DOP — it is a property of the complete system: plasticizer, stabilizer class, ratio, co-stabilizers, and processing conditions acting together. Ca/Zn demands lubricant rebalancing and precise calcium-to-zinc ratios. Organotin tolerates DOP readily but reaches peak performance through relay blending with Ca/Zn at a 5:4:1 ratio. Both classes require controlled mixer discharge temperatures to convert formulated stabilizer into available stabilizer.
The next step for any formulator dealing with DOP-stabilizer compatibility issues: run a Congo red test at your actual processing temperature, not the textbook 180 C. Your thermal stability numbers under real conditions will tell you whether you have a formulation problem or a dispersion problem — and those two problems have completely different solutions.